2X Match My Gift
This National Cancer Survivors Month, your gift is matched to expand treatment options and bring more patients into survivorship through Project Cure CRC.


Minimally invasive surgery (MIS) refers to laparoscopic surgery and robotic surgery. These two types of surgery are different based on the tools used during the procedure. The surgeon will either use robotics or laparoscope tools. Surgeons will usually recommend a laparoscopic or robotic procedure based on their own experience and preference.
Minimally invasive surgeries are much more common now because this type of surgery can offer colorectal cancer patients a less painful way to remove part of the colon or the entire colon, create an ostomy, or reconnect and reconstruct the colon.
Minimally invasive surgery can also be used, in some cases, to remove rectal tumors.
Many studies have shown that these surgeries are just as safe as open surgery but they make recovery faster and easier.
Most patients can undergo minimally invasive surgery, such as those with:
Open surgery might still be recommended for patients with:
Talk to your doctor to find out if minimally invasive surgery is an option for you.

For many cancer patients, the end of active treatment brings a new kind of dread. Scans every few months and anxious waits for results. A blood test is changing that experience for a growing number of patients.
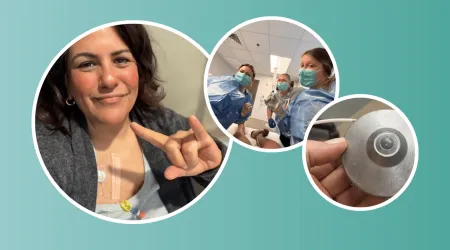
When Helen was diagnosed with metastatic colorectal cancer, the biggest concern for her medical team was that her cancer had spread to her liver. One type of therapy, hepatic artery infusion, offered a path forward, but there was a catch. Nobody at her hospital had ever done it before. She'd have to be first.

hrough Project Cure CRC, the Alliance is fueling bold, early-stage research with the potential to transform colorectal cancer treatment. Dr. Lisa Mielke’s groundbreaking work explores how the gut’s immune system and nerve signaling influence cancer growth—opening the door to new therapeutic approaches, including repurposed existing drugs. This is what’s possible when promising ideas get the support they need to move forward.