2X Match My Gift
This National Cancer Survivors Month, your gift is matched to expand treatment options and bring more patients into survivorship through Project Cure CRC.


Michelle Cappel owes a lot to biomarker testing — seven years of life and counting.
“I shouldn’t be alive, and I’m well aware of that,” she says. “But I’m here to use my voice and help other people.”
Michelle was diagnosed in 2017 with stage IV (metastatic) colorectal cancer. After diagnosis, Michelle’s first oncologist told her that she had less than a year to live.
Devastated and desperate for a future, Michelle received a second opinion from MD Anderson Cancer Center in Houston.
Unlike her first oncologist, MD Anderson’s doctors used biomarker testing to identify the unique characteristics of Michelle’s tumor and possibly find a treatment that could add years to her life.
Testing showed that Michelle’s tumor had biomarkers that could respond well to treatment using the drug Folfox, which she began immediately.
Michelle’s tumors shrank. With surgery lending a hand, too, she was declared NED (no evidence of disease). She stayed disease-free for nearly three years.
What is biomarker testing?
Biomarker testing provides detailed information about a patient’s tumor. A doctor can examine test results to identify specific biomarkers and their levels, which can inform treatment decisions. They can also help determine eligibility for clinical trials.
Biomarker testing is useful in both early and advanced (metastatic) stages of colorectal cancer. For patients with advanced cancer, biomarker testing is used to learn whether cancer has any targetable characteristics. If it does, targeted therapy or immunotherapy may be a treatment option.
Anyone who is diagnosed with colorectal cancer, regardless of stage, should be tested for high microsatellite instability (MSI-H). Patients who are diagnosed with stage lV (metastatic) colon or rectal cancer should be tested for at least four predictive biomarkers: KRAS, NRAS, BRAF, and HER2.
A different route
In 2021, Michelle started to experience leg pain. A scan showed a large metastasis on her femoral nerve, which runs from the pelvis to the front of the leg, above the knee. Treatment threatened paralysis.
“I was extremely blessed and lucky when they were in surgery and realized it was not on the nerve but in the muscle,” Michelle says. The surgeon could cut out the cancer and save enough muscle for use of the leg. “I am proof it doesn’t always go the way they think it will go.”
Combined with pharmaceutical treatment and radiation, Michelle eventually returned to NED status.
Unfortunately, a second recurrence was found in June 2023. A scan revealed peritoneal metastasis, which is often treated with Hyperthermic Intraperitoneal Chemotherapy (HIPEC) — a major, invasive surgery with a long recovery time.
Rather than go that route, Michelle is watching social media, websites, and clinical trial finders to keep tabs on new science, hopeful that a trial for her particular biomarkers and situation will become available.
“For new patients, it’s imperative to get the best care, and if no one has mentioned biomarker testing, they need to find their voice and ask their doctors about it,” Michelle says. “It will help save their lives.”

For many cancer patients, the end of active treatment brings a new kind of dread. Scans every few months and anxious waits for results. A blood test is changing that experience for a growing number of patients.
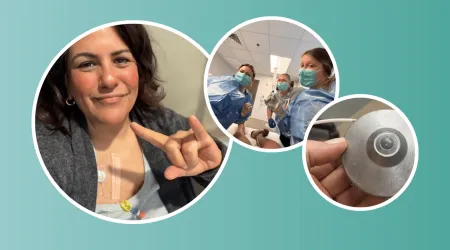
When Helen was diagnosed with metastatic colorectal cancer, the biggest concern for her medical team was that her cancer had spread to her liver. One type of therapy, hepatic artery infusion, offered a path forward, but there was a catch. Nobody at her hospital had ever done it before. She'd have to be first.
Understanding colon cancer sidedness is one more tool to help patients ask the right questions and advocate for the most effective treatment plan.