2X Match My Gift
This National Cancer Survivors Month, your gift is matched to expand treatment options and bring more patients into survivorship through Project Cure CRC.
Many colorectal cancer patients will have a temporary ostomy after surgery but less than 10% of patients will end up having a permanent ostomy.

Think of your ostomy’s function as you did your natural bowel movements. You still have the same bowel; it just might be redirected for a little while. The real change is having stool come out of an opening made on your stomach.

Before your ostomy surgery, you and your surgeon will discuss the best place for your stoma. They will examine your abdomen will you are in different positions – sitting, standing, or lying down.
The stoma is usually placed below the waistline, in the lower part of the abdomen.
If you have obesity, the stoma may be placed above the waistline.
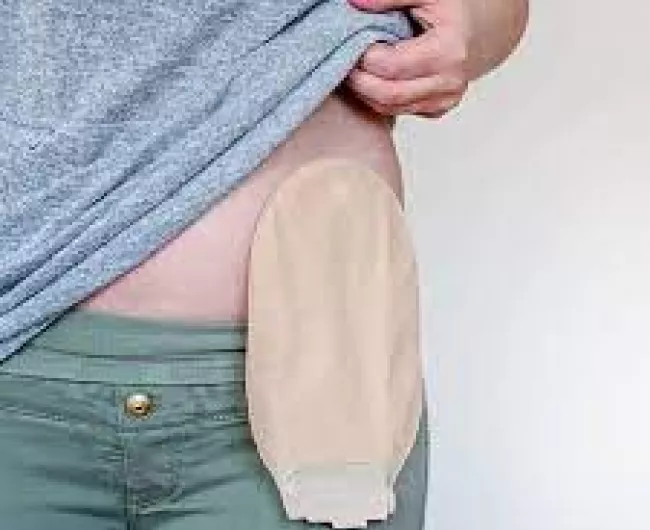
There are several factors that affect where your stoma is placed. Your surgeon will place it in location that:
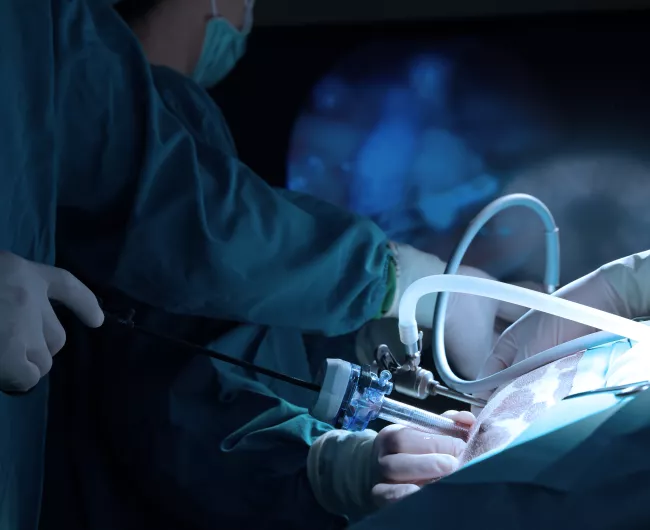
Your ostomy is often created by your surgeon during a colectomy. You will be under general anesthesia, and the surgery may a laparoscopic,robotic-assisted, or an open procedure.
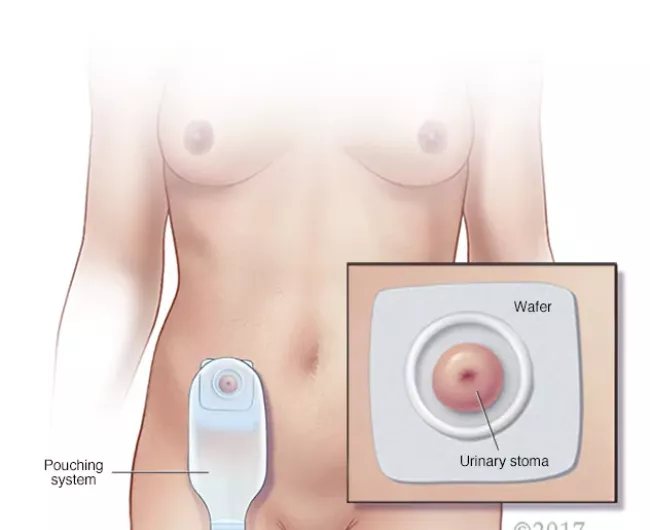
Most people recover from ostomy surgery with few difficulties.
You will have a stoma visible on your abdomen. The stoma looks like a small, pink circle about the size of a quarter. It may be flat against your body or protrude out a bit. The stoma will be connected to a pouch that has an opening where it can be emptied.
You may notice some changes in your digestive system after your stoma surgery:
As your digestive tract heals, these symptoms should improve or lessen over time. Be sure to talk to your doctor about any changes you notice and let them know if these changes are troublesome.
As with any surgery, complications are rare but may arise. In addition to infection and bleeding, there are some specific complications that can occur after a stoma is placed.
Contact your doctor right away if you notice any of the following:
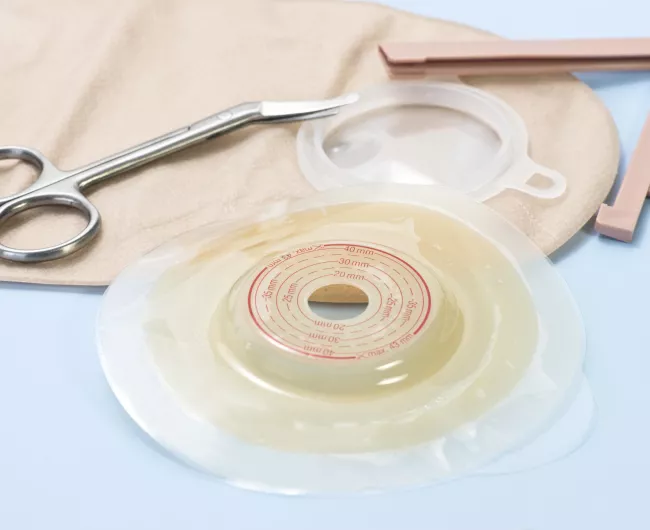
An ostomy nurse or other specialist will teach you how to manage the care of your new ostomy.
Before your surgery, ask your surgeon who will be teaching you.

For many cancer patients, the end of active treatment brings a new kind of dread. Scans every few months and anxious waits for results. A blood test is changing that experience for a growing number of patients.
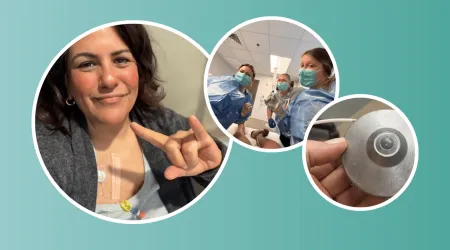
When Helen was diagnosed with metastatic colorectal cancer, the biggest concern for her medical team was that her cancer had spread to her liver. One type of therapy, hepatic artery infusion, offered a path forward, but there was a catch. Nobody at her hospital had ever done it before. She'd have to be first.

hrough Project Cure CRC, the Alliance is fueling bold, early-stage research with the potential to transform colorectal cancer treatment. Dr. Lisa Mielke’s groundbreaking work explores how the gut’s immune system and nerve signaling influence cancer growth—opening the door to new therapeutic approaches, including repurposed existing drugs. This is what’s possible when promising ideas get the support they need to move forward.