2X Match My Gift
This National Cancer Survivors Month, your gift is matched to expand treatment options and bring more patients into survivorship through Project Cure CRC.
You might have seen in the news: this week, the FDA approved the first generic version of capecitabine (Xeloda to most of us), an oral chemotherapy pill used to treat breast and colon cancers.
So what does this mean? Basically, when generic versions of drugs become available to the masses, it can mean better access for patients. We all know – brand-name drugs can be VERY pricey, sometimes so much so that people who need the drug can’t even afford it. The good news: typically, generic forms of drugs cost significantly less than the brand-name equivalents and still provide the same effectiveness.
So in this case, patients who had been receiving the brand-name version of Xeloda will be spending less for the same treatment and patients who couldn’t afford the medication can now ask their doctors if the generic version is right for them.
Different Drug, Same Quality
If you’re worried about the quality of these new, non-name-brand forms of Xeloda, don’t be. Remember that all FDA-approved generic drugs are subject to the same stringent guidelines and are held to the same standards as their brand-name equivalent. Actually, as cancer continues to be treated as a chronic illness, we will likely be seeing other generic versions of popular treatments become available. A generic drug may look or taste different or even have different inactive ingredients, but in general, it shouldn’t be any less effective for you.
Ow, Ow! Choose Me! Over Here!
So then why are generic drugs SO much cheaper? Because the companies that offer these drugs didn’t have to spend the money to research, develop and market them (and as you can imagine – this process this isn’t a cheap one.) Another reason: more forms of a drug on the market means increased competition; companies are forced to keep their prices lower so that people will actually buy their product.
There you have it! If you have questions, call our Toll-free Helpline at (877) 422-2030. Find more information here.

The Colorectal Cancer Alliance is urging Americans to prioritize colorectal cancer screening, as the American Cancer Society (ACS) released updated guidelines today.

For many cancer patients, the end of active treatment brings a new kind of dread. Scans every few months and anxious waits for results. A blood test is changing that experience for a growing number of patients.
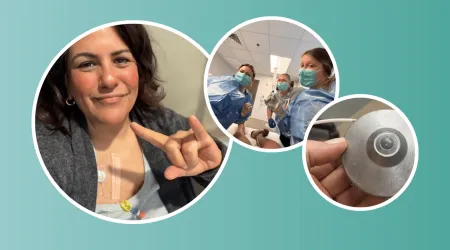
When Helen was diagnosed with metastatic colorectal cancer, the biggest concern for her medical team was that her cancer had spread to her liver. One type of therapy, hepatic artery infusion, offered a path forward, but there was a catch. Nobody at her hospital had ever done it before. She'd have to be first.