2X Match My Gift
This National Cancer Survivors Month, your gift is matched to expand treatment options and bring more patients into survivorship through Project Cure CRC.
October 26, 2017 (WASHINGTON)–VARUBI, a highly anticipated treatment for colorectal patients who suffer from delayed chemotherapy-induced nausea, has been approved by the Food and Drug Administration for delivery via one-time intravenous administration. This new way to administer VARUBI provides healthcare providers with flexibility to treat patients in a more individualized manner.
VARUBI addresses delayed nausea that can arrive 24 to 120 hours after the start of chemotherapy. If nausea is not managed aggressively, it can lead to weight loss, dehydration, and hospitalization. Quality-of-life issues are incredibly important for patients and families dealing with colorectal cancer and chemotherapy. The approval of VARUBI is good step forward in addresses some of these concerns.
For more information about VARUBI, please access:
https://globenewswire.com/news-release/2017/10/25/1153687/0/en/TESARO-Announces-U-S-FDA-Approval-of-VARUBI-IV-for-Delayed-Nausea-and-Vomiting-Associated-With-Cancer-Chemotherapy.html

A first-of-its-kind trial platform in the colorectal cancer space, Project Cure CRC’s KLEOS is being driven by the Colorectal Cancer Alliance, the leading nonprofit dedicated to ending the disease, in collaboration with GCAR, a pioneer in the design and implementation of innovative clinical trials.

For many cancer patients, the end of active treatment brings a new kind of dread. Scans every few months and anxious waits for results. A blood test is changing that experience for a growing number of patients.
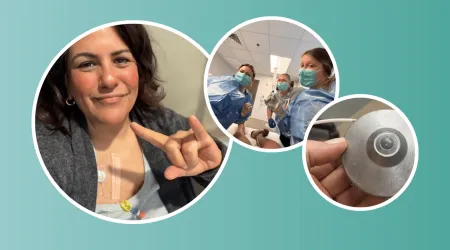
When Helen was diagnosed with metastatic colorectal cancer, the biggest concern for her medical team was that her cancer had spread to her liver. One type of therapy, hepatic artery infusion, offered a path forward, but there was a catch. Nobody at her hospital had ever done it before. She'd have to be first.